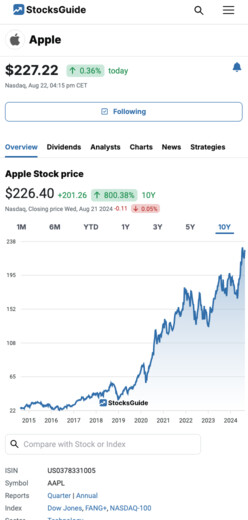
Madrigal Pharmaceuticals, Inc. Stock News
Madrigal Pharmaceuticals, Inc. Stock News
$212.43
0.69%
yesterday
Nasdaq,
Sep 27, 10:00 pm CET
Madrigal Pharmaceuticals, Inc. Stock News
Neutral
InvestorPlace
●
2 months ago
Initially, I had every intention to write a cookie-cutter assessment of intriguing biotech stocks; that is, I wanted to say that enterprises within this specialized healthcare field will always be relevant as humans leverage advanced innovations to address vexing conditions and diseases. That remains true.
Neutral
The Motley Fool
●
3 months ago
Madrigal Pharmaceutical is the only biotech in its market. Its market is likely to be a fairly large one.
Negative
The Motley Fool
●
3 months ago
Madrigal Pharmaceuticals earned an important regulatory approval this year. However, the biotech faces risks, including the need for a post-marketing study.
Neutral
The Motley Fool
●
3 months ago
Madrigal Pharmaceuticals is the only company with an approved drug for MASH. Eli Lilly's latest clinical trial data shows that its candidate may perform even better.
Positive
The Motley Fool
●
3 months ago
Madrigal Pharmaceuticals currently has undisputed dominance in its main market. There are powerful actors trying to elbow their way in and change that status.
Neutral
Seeking Alpha
●
4 months ago
Madrigal Pharmaceuticals, Inc. stock has enjoyed peaks and troughs as it has pursued its path to the approval of NASH drug Rezdiffra. The company's stock was buoyant yesterday, enjoying a 15% gain after Wolfe Research initiated positive coverage. Competition from GLP-1 agonists like Eli Lilly's tirzepatide and Novo Nordisk's semaglutide are expected to pose a threat to Rezdiffra's commercial su...
Neutral
The Motley Fool
●
4 months ago
Madrigal Pharmaceuticals has a monopoly in its main market -- for the moment. Viking Therapeutics has a good candidate, but it could face powerful competitors.
Neutral
GlobeNewsWire
●
4 months ago
CONSHOHOCKEN, Pa., June 06, 2024 (GLOBE NEWSWIRE) -- Madrigal Pharmaceuticals, Inc. (NASDAQ:MDGL), a biopharmaceutical company focused on delivering novel therapeutics for nonalcoholic steatohepatitis (NASH), today announced results from new analyses of the Phase 3 MAESTRO-NASH trial of Rezdiffra being presented at the EASL Congress, which takes place from June 5-8, 2024 in Milan, Italy.
Register for Free
StocksGuide is the ultimate tool for easily finding, analyzing and tracking stocks. Learn from successful investors and make informed investment decisions. We empower you to become a confident, independent investor.