Takeda Pharmaceutical Co. Ltd. Sponsored ADR Stock News
Takeda Pharmaceutical Co. Ltd. Sponsored ADR Stock News
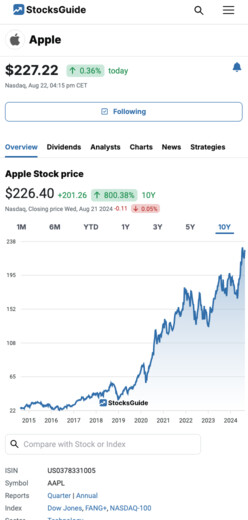
$14.54
0.75%
yesterday
NYSE,
Sep 27, 10:10 pm CET
Takeda Pharmaceutical Co. Ltd. Sponsored ADR Stock News
Positive
InvestorPlace
●
2 months ago
The biotechnology sector doesn't need to rizz up investors, as the kids like to say. Unless the human desire to find solutions for vexing conditions and diseases somehow goes away in a dystopian paradigm, the ecosystem is safe.
Positive
Seeking Alpha
●
2 months ago
AC Immune SA's Takeda Pharmaceutical Company partnership brought in an upfront payment of $100 million, with the potential to earn up to $2.1 billion in milestone payments. Interim safety and immunogenicity data from the phase 2 ABATE trial using ACI-24.060 for the treatment of Down Syndrome patients with AD, expected 2nd half of 2024. The global Alzheimer's therapeutics market size is expected...
Positive
Seeking Alpha
●
2 months ago
Takeda Pharmaceutical Company Limited showed signs of improvement despite revenue declines in oncology and neurosciences, thanks to gains in gastrointestinal and inflammation sectors and strategic leadership changes. The company's “returning to growth program” aims to increase operating margins by 100-250 basis points annually from 2025 onwards. Despite a challenging FY2023 with a significant n...
Neutral
Seeking Alpha
●
3 months ago
The small biotech sector has badly underperformed the major market indices so far in 2024.
Neutral
Business Wire
●
3 months ago
OSAKA, Japan--(BUSINESS WIRE)--Takeda (TOKYO:4502/NYSE:TAK) has announced new assignments of directors, determined at the Board of Directors meeting and at the Audit and Supervisory Committee meeting, following the 148th Ordinary Meeting of Shareholders, held in Osaka today. Takeda's Board of Directors has 11 members serving as external directors out of a total of 14 members, helping to ensure ...
Neutral
Business Wire
●
3 months ago
OSAKA, Japan & CAMBRIDGE, Mass.--(BUSINESS WIRE)--Takeda (TSE:4502/NYSE:TAK) today announced that LIVTENCITY® (maribavir) has been approved by the Japanese Ministry of Health, Labour and Welfare (MHLW) for post-transplant cytomegalovirus (CMV) infection/disease that is refractory to existing anti-CMV therapies.4 LIVTENCITY is the first and only post-transplant anti-CMV treatment approved in Jap...
Neutral
Business Wire
●
3 months ago
OSAKA, Japan & CAMBRIDGE, Mass.--(BUSINESS WIRE)--Takeda (TSE:4502/NYSE:TAK) today presented positive results from its Phase 2b, randomized, double-blind, placebo-controlled study evaluating the safety, tolerability and efficacy of mezagitamab (TAK-079) in patients with persistent or chronic primary immune thrombocytopenia (ITP), a rare immune-mediated bleeding disorder. ITP is characterized by...
Neutral
Business Wire
●
3 months ago
OSAKA, Japan & CAMBRIDGE, Mass.--(BUSINESS WIRE)--Takeda (TSE:4502/NYSE:TAK) today announced that the European Commission (EC) approved FRUZAQLA (fruquintinib) as a monotherapy indicated for the treatment of adult patients with metastatic colorectal cancer (mCRC) who have been previously treated with available standard therapies, including fluoropyrimidine-, oxaliplatin-, and irinotecan-based c...
Register for Free
StocksGuide is the ultimate tool for easily finding, analyzing and tracking stocks. Learn from successful investors and make informed investment decisions. We empower you to become a confident, independent investor.